/periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
Isotopes are never isolated in the periodic table they are constantly gathered together under a solitary element. For instance, carbon has three normally happening isotopes: the majority of its particles have six protons and most have six neutrons also, however around one percent has seven neutrons, and an extremely little portion has eight neutrons. Most elements have contrasting quantities of neutrons among various atoms, with these variations being alluded to as isotopes. Variously manufactured radionuclides of naturally occurring elements have likewise been created in various research centers.Įvery chemical element has a unique atomic number (Z) that represents the number of protons in its nucleus. 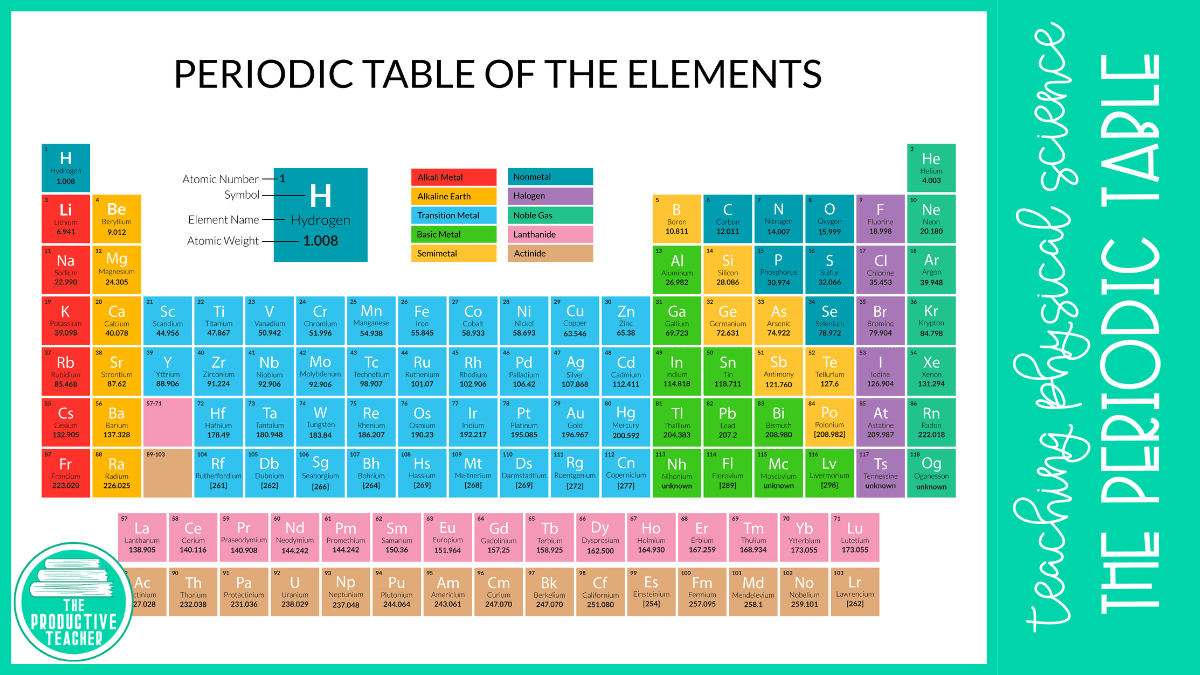
The amalgamation of elements having higher atomic numbers is as of now being pursued/analyzed: these elements would start the eighth line, and hypothetical work has been done to recommend conceivable possibilities for this augmentation. 
Elements 95 to 118 have been completely created and developed in research centers or atomic reactors. The initial 94 elements are all naturally occurring elements, however, some are discovered just in trace or scarce amounts and a couple of them were found in nature simply in the wake of having originally been synthesized. The elements from atomic numbers 1 (hydrogen) through 118 (oganesson) have been found or incorporated, finishing seven full lines of the periodic table. Additionally, shown are four basic rectangular zones or blocks related to the filling of various atomic orbitals. Six groups have acknowledged names just as appointed numbers: for instance, group 17 elements are the halogen group and group 18 are the noble gasses group. The segments, called groups, contain elements with approximately the same chemical behavior. The seven lines of the table, called periods, by and large, have metals on the left and nonmetals on the right side. The structure of the table shows the periodic patterns. (In the modern periodic table, a group or family corresponds to one vertical column.A tabular platform of the chemical elements in the periodic table which is also called the periodic table of elements is organized by the atomic number, electron setup, and persistent compound properties. The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. 
There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
